My research involves the use of biochemical, biophysical and structural biology strategies to elucidate the structure and function of proteins which will solve some of the biological problems. I study on three major issues. The first is structural and functional study of MDC1-FHA domain in DNA damage response. The second is structural study of Fha interaction with phosphothreonine of TssL activates type VI section in Agrobacterium tumefaciens. The third is structural, biochemical, and spectra study of McHr and MDH in Methylococcus capsulatus (Bath).
Structural and Functional Study of MDC1-FHA domain in DNA Damage Response
|
|
My research involves the use of biochemical, biophysical and structural biology strategies to elucidate the structure and function of proteins in response to DNA damage which will solve some of the biological problems. The proteins I study are mediator of DNA damage checkpoint 1 (MDC1) and checkpoint kinase 2 (CHK2).
The MDC1 functions as a key mediator that interacts with multiple proteins involved in the DNA damage response pathway (DDR). The FHA domain of MDC1 (MDC1-FHA) plays critical but elusive role in the regulation of DDR. Literatures reported that it binds to not only sensor proteins including ATM and MRN complex but also to effector protein such as CHK2. By which a complicated phospho-signaling network, MDC1 controls the sensing, initiating, and mediating steps that lead to downstream repair pathways, cell-cycle checkpoint, and apoptosis. MDC1 therefore is responsible for the maintenance of genomic stability and prevention of tumorigenesis. |
|
Previously, we published the crystal structures of mouse MDC1-FHA and its complex with a human CHK2 peptide containing pThr68. Strikingly, we and the other two groups independently found that, unlike most of known FHA domains, MDC1-FHA exists as an intrinsic dimer in solution and in crystals. We further pursue to validate the biological relevance of such FHA-directed dimerization. First we select different MDC1-FHA mutants that disrupt the dimerization while maintain pThr-binding ability. The ectopically expressed full-length MDC1 protein harboring such mutation not only failed to dimerize in vivo as suggested by self-association ability using immunoprecipitation and split-GFP experiments, but also failed to rescue cellular radio-sensitivity caused by MDC1 silencing.
|
|
|
Interestingly, the mutated MDC1 proteins exhibit much faster and intensified recruitment mobility in response to DNA double-strand breaks (DSBs) created by in vivo laser micro-irradiation. Similar elevated mobility of MDC1 protein can also be seen when treating the cells with proteasome inhibitor and AKT inhibitor, suggesting that the promoted MDC1 recruitment by dimer-disruption is likely due to decelerated protein turnover on DNA lesions. It has been reported that MDC1 is degraded through SUMOylation-regulated ubiquitination in response to DSBs. We therefore hypothesize that the dimerization is a prerequisite for SUMOylation and ubiqutination of MDC1 and is essential for subtle MDC1 protein turnover on DSBs sites by which the accurate DNA damage signal can be delivered. We next will apply detailed molecular and cellular analysis to decipher the functional significance of dimeric MDC1-FHA in the maintenance of genomic stability.
|
Structural, Biochemical, and Spectra Study of McHr and MDH in Methylococcus capsulatus (Bath)
|
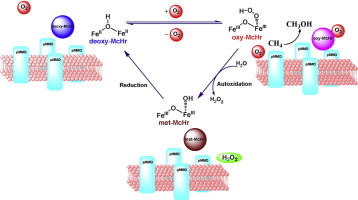
Recently, a native bacteriohemerythrin (McHr) has been identified in Methylococcus capsulatus (Bath). Both the particulate methane monooxygenase (pMMO) and McHr are over-expressed in cells of this bacterium when this strain of methanotroph is cultured and grown under high copper to biomass conditions. It has been suggested that the role of the McHr is to provide a shuttle to transport dioxygen from the cytoplasm of the cell to the intra-cytoplasmic membranes for consumption by the pMMO. Indeed, McHr enhances the activity of the pMMO when pMMO-enriched membranes are used to assay the enzyme activity. We find that McHr can dramatically improve the activity of pMMO toward the epoxidation of propylene to propylene oxide. The maximum activity is observed at a pMMO to McHr concentration ratio of 4:1, where we have obtained specific activities of 103.7 nmol propylene oxide/min/mg protein and 122.8 nmol propylene oxide/min/mg protein at 45 °C when the turnover is driven by NADH and duroquinol, respectively. These results are consistent with the suggestion that the bacterium requires McHr to deliver dioxygen to the pMMO in the intra-cytoplasmic membranes to accomplish efficient catalysis of methane oxidation when the enzyme is over-expressed in the cells.
|
|
|
The bacteriohemerythrin (McHr) from Methylococcus capsulatus (Bath) is an oxygen carrier that serves as a transporter to deliver O2 from the cytosol of the bacterial cell body to the particulate methane monooxygenase residing in the intracytoplasmic membranes for methane oxidation. More recently, we report X-ray protein crystal structures of the recombinant wild type (WT) McHr and its L114A, L114Y and L114F mutants. The structure of the WT reveals a possible water tunnel in the McHr that might be linked to its faster autoxidation relative to hemerythrin in marine invertebrates. With Leu114 positioned at the end of this putative water tunnel, the hydrophobic side chain of this residue seems to play a prominent role in controlling the access of the water molecule required for autoxidation. This hypothesis is examined by comparing the autoxidation rates of the WT McHr with those of the L114A, L114Y and L114F mutants. The biochemical data are correlated with structural insights derived from the analysis of the putative water tunnels in the various McHr proteins provided by the X-ray structures.
|
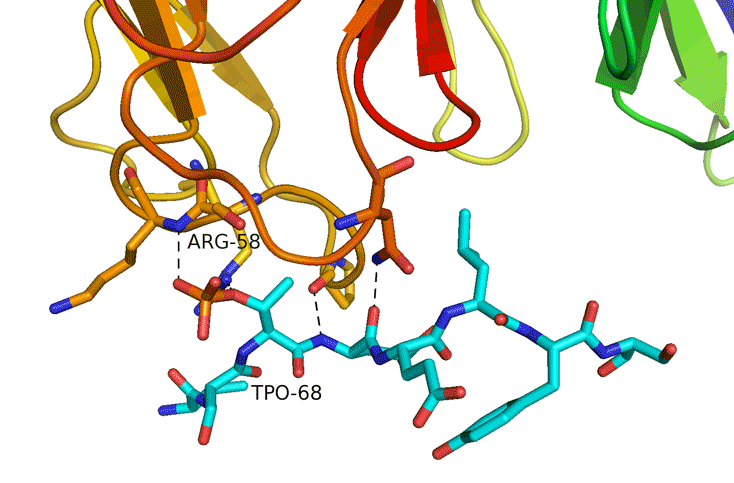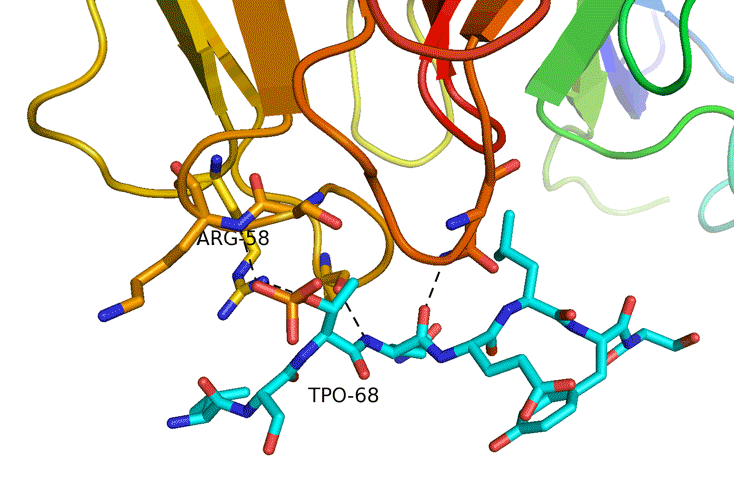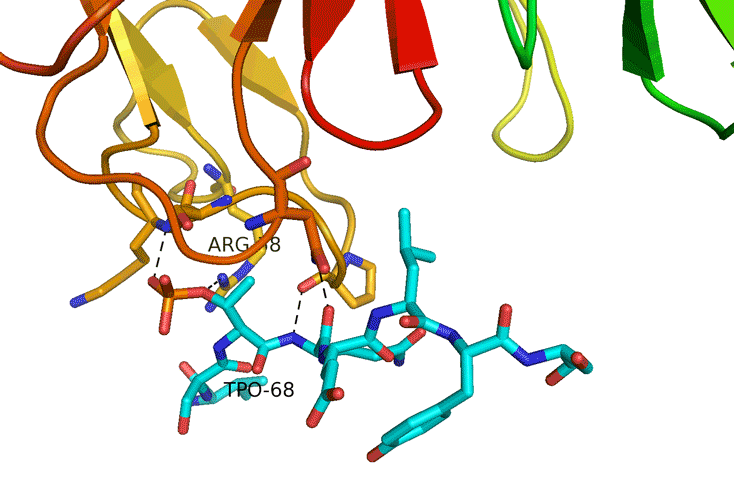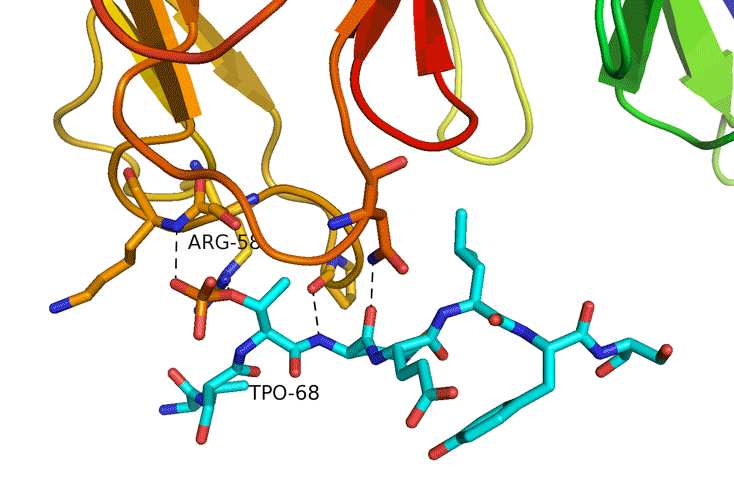
Structural Study of Fha Interaction with Phosphothreonine of TssL Activates type VI section in Agrobacterium tumefaciens
|
|
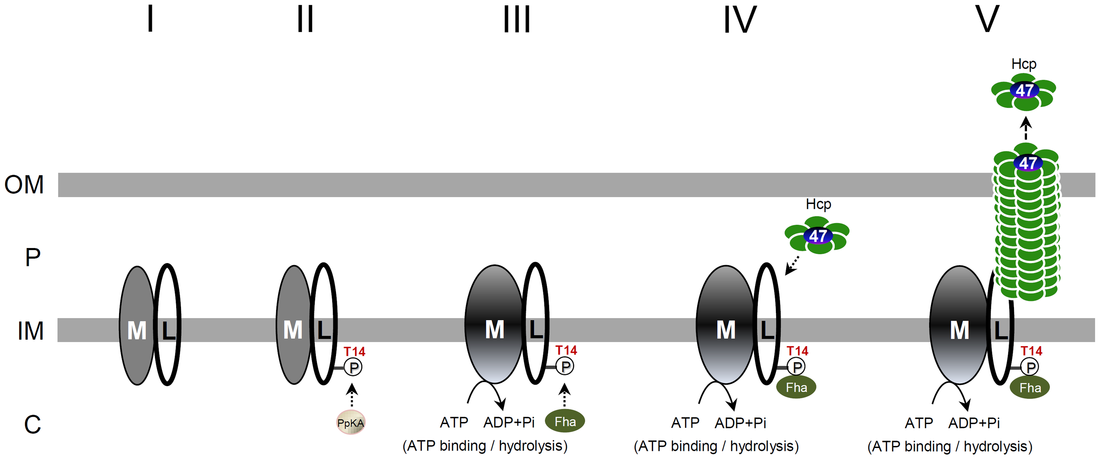
The type VI secretion system (T6SS) is a widespread protein secretion system found in many Gram-negative bacteria. T6SSs are highly regulated by various regulatory systems at multiple levels, including post-translational regulation via threonine (Thr) phosphorylation. The Ser/Thr protein kinase PpkA is responsible for this Thr phosphorylation regulation, and the forkhead-associated (FHA) domain-containing Fha-family protein is the sole T6SS phosphorylation substrate identified to date. Here we discovered that TssL, the T6SS inner-membrane core component, is phosphorylated and the phosphorylated TssL (p-TssL) activates type VI subassembly and secretion in a plant pathogenic bacterium, Agrobacterium tumefaciens. Combining genetic and biochemical approaches, we demonstrate that TssL is phosphorylated at Thr 14 in a PpkA-dependent manner. Further analysis revealed that the PpkA kinase activity is responsible for the Thr 14 phosphorylation, which is critical for the secretion of the T6SS hallmark protein Hcp and the putative toxin effector Atu4347. TssL phosphorylation is not required for the formation of the TssM-TssL inner-membrane complex but is critical for TssM conformational change and binding to Hcp and Atu4347. Importantly, Fha specifically interacts with phosphothreonine of TssL via its pThr-binding motif in vivo and in vitro and this interaction is crucial for TssL interaction with Hcp and Atu4347 and activation of type VI secretion. In contrast, pThr-binding ability of Fha is dispensable for TssM structural transition. In conclusion, we discover a novel Thr phosphorylation event, in which PpkA phosphorylates TssL to activate type VI secretion via its direct binding to Fha in A. tumefaciens. A model depicting an ordered TssL phosphorylation-induced T6SS assembly pathway is proposed.
|